PSE-Lab has just published a new paper on “Computers and Chemical Engineering” titled:
Definition and validation of a patient-individualized physiologically based pharmacokinetic model
Roberto Andrea Abbiati, Gaetano Lamberti, Mario Grassi, Francesco Trotta, Davide Manca
Abstract
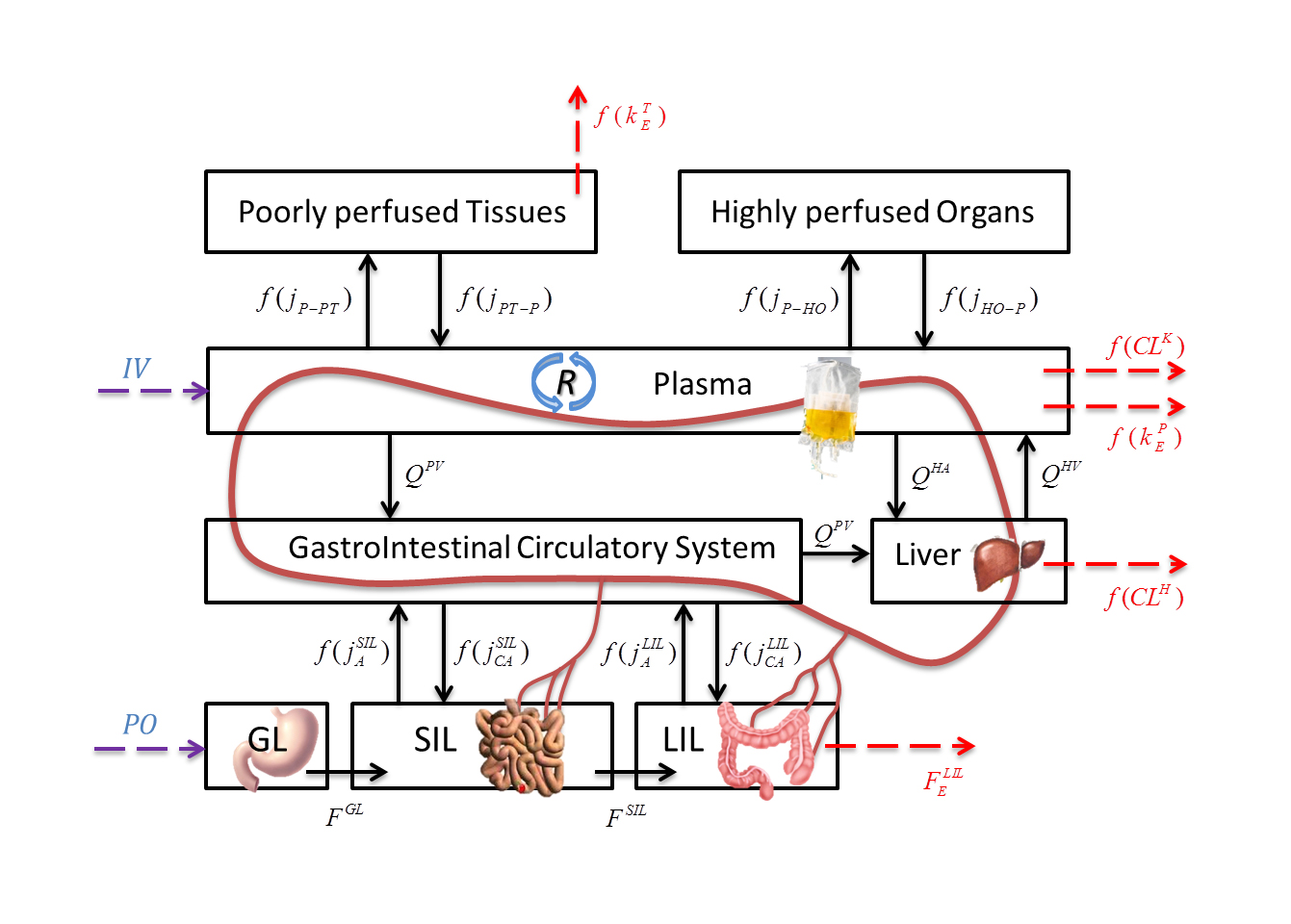
Pharmacokinetic modeling based on a mechanistic approach is a promising tool for drug concentration prediction in living beings. The development of a reduced physiologically based pharmacokinetic model (PBPK model), is performed by lumping organs and tissues with comparable characteristics respect to drug distribution phenomena. The proposed reduced model comprises eight differential equations and 18 adaptive parameters. To improve the quality of the PBPK model these adaptive parameters are alternatively: (i) individualized according to literature correlations on the physiological features of each patient; (ii) assigned as constants based on the features of either human body or drug properties; (iii) regressed respect to experimental data.
The model predictive capability is validated with experimental blood concentrations of remifentanil, an analgesic drug, administered via bolus injection with four doses (2, 5, 15, 30 µg/kg) to mixed groups of patients. Concentration profiles for the four simulated doses reveal a rather good consistency with experimental data.
Highlights
- A pharmacokinetic (PK) model describes the drug biodistribution in the human body
- The PK model is based on human anatomy and physiology
- Some model parameters are personalized according to specific features of patients
- Case-study: PK of remifentanil analgesic with concentrations in organs and tissues
Interested readers can download a copy of the paper.
To cite the paper, please use this reference:
Roberto Andrea Abbiati, Gaetano Lamberti, Mario Grassi, Francesco Trotta, Davide Manca, DEFINITION AND VALIDATION OF A PATIENT-INDIVIDUALIZED PHYSIOLOGICALLY BASED PHARMACOKINETIC MODEL, Computers & Chemical Engineering, 84, 394–408, (2016)
http://dx.doi.org/10.1016/j.compchemeng.2015.09.018