PSE-Lab published a new article on Industrial and Engineering Chemistry Research:
Enterohepatic Circulation Effect in Physiologically Based Pharmacokinetic Models: The Sorafenib Case
Roberto Andrea Abbiati, Davide Manca
Abstract
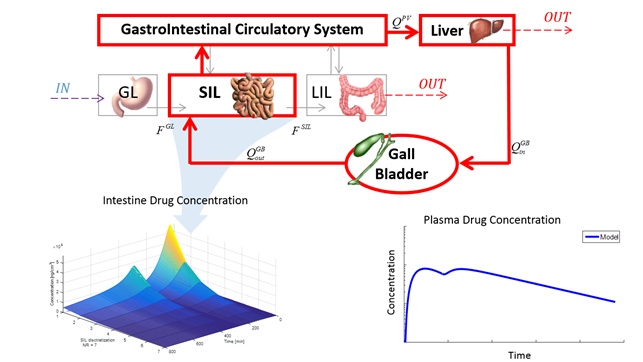
Sorafenib was recently approved by both FDA and EMA for the treatment of unresectable hepatocellular carcinoma (HCC), renal cell carcinoma (RCC), and thyroid cancer (DTC). This paper proposes a physiologically based model to describe its pharmacokinetic properties. Special attention is devoted to the enterohepatic circulation, which causes the characteristic double‑peak in the drug’s concentration-time curves.
This physiologically‑based pharmacokinetic (PBPK) model has a structure suitable for different mammals. The paper focuses on the anatomy and physiology of mice aiming to develop a physiologically consistent model to produce accurate simulation of sorafenib pharmacokinetics, in agreement with literature data. The in silico description of sorafenib pharmacokinetics allows understanding further its properties, by complementing the experimental data with numerical simulations based on a mechanistic approach. Modeling and simulation is a rapidly growing branch of quantitative systems pharmacology, and supports new drug development by shortening analysis time, reducing costs, and delivering reliable pharmacokinetic knowledge.
Keywords
PBPK models; Model lumping; Enterohepatic circulation; Biodistribution; Hepatocellular carcinoma; Sorafenib pharmacokinetics; Quantitative Systems Pharmacology.